As COVID Vaccines Seem More Plausible, Oregon Focuses on Its Distribution Plan

As potential coronavirus vaccines continue to prove effective in research trials, Oregon is prepping its distribution plan.
Image: Brian Breneman
With promising news of potential coronavirus vaccines produced by Pfizer and Moderna, both of which have been shown to be more than 90 percent effective in treating patients with COVID-19, Oregon’s coronavirus vaccine distribution plan is in the spotlight.
Joe Sullivan, senior health advisor for the Oregon Health Authority’s COVID-19 response, says things are moving faster than expected and “puts urgency” on coordinating an airtight vaccination plan to set into motion if and when a vaccine is available to the public, which he says could be as early as mid-December.
“While the numbers of 90-plus percent are still interim, if those numbers hold, as we hope that they will, that bodes very well for having an effective vaccine that, when wisely deployed, will really bring the numbers in Oregon down and will allow us to get back to Portland and Oregon as we love it,” Sullivan says.
Oregon’s 136-page COVID-19 vaccination plan, which OHA sent to the Centers for Disease Control in October, lays out a three-phase framework, per federal guidance, that focuses on health equity and the pandemic’s disproportionate effects on tribal communities and communities of color.
So the big question: Who gets the vaccine first? According to the vaccination plan, healthcare personnel, including first responders and those who work in hospitals and long-term care facilities, would likely receive first dibs. After that, three groups are next: those over 65, those with chronic health conditions (obesity, hypertension, etc.) that increase their risk of developing COVID symptoms, and essential workers. Phase 2 would focus on those groups disproportionately affected by the coronavirus, and Phase 3, a “period of discovery,” says Sullivan, would focus on the general public.
Only a draft, the vaccine guidelines are likely to change, but Sullivan says early challenges include reaching rural Oregon communities and creating a “cold chain” to keep the vaccines at ultra-cold temperatures during distribution. Of course, vaccines alone won’t curb the recent spike in Oregon and elsewhere across the country. But ensuring everyone is vaccinated will, Sullivan says, which is perhaps more difficult than it sounds. An NPR/PBS NewsHour/Marist poll in August found only about 60 percent of Americans say they’d be willing to get the vaccine.
Sullivan says there is always a group that is suspicious of vaccines, and with the COVID vaccine’s accelerated timeline “there are more fence-sitters than usual.”
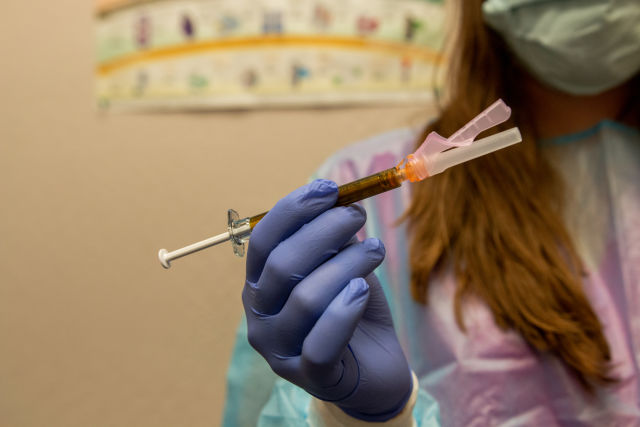
A study coordinator at the Clinical Research Institute of Southern Oregon holds a dosage of the Moderna vaccine used in recent trials. The Moderna vaccine has shown almost 95 percent effectiveness in treating patients who have contracted the coronavirus.
Image: Gabriel Granillo
Operation Warp Speed, under which the Moderna, Pfizer, and other coronavirus vaccines are being produced, has accelerated the time it typically takes to produce treatments or vaccines, compressing what usually takes years into a few months. But health experts say trials have increased in size to accommodate for the increased timeline and that the normal safeguards are still in place and being followed. The question of when or if revaccination is needed will be determined as a potential vaccine is doled out and administered to the general public.
“It’s also possible that we will learn things from the first vaccines that are approved that will allow us to tweak them and make them more effective later,” Gregg Lucksinger says.
Lucksinger is the medical director at the Clinical Research Institute of Southern Oregon, which recently conducted research for Moderna’s coronavirus vaccine.
“Ideally we’ll see a couple of dozen candidates get to human trials and a bunch of those will be shown to be safe and effective because we need to vaccinate over seven billion people. It’s a big production challenge. It’s a big logistics challenge. It may be that some of the vaccines work great in adults but don’t work that good in young children, or work great in young children but not in the elderly,” Lucksinger says. “I think there is a reasonable optimism that we’ll have several approved in the next year or two.”
Even with a vaccine or vaccines on the horizon, Sullivan says “the next couple of months are going to be dark.” Recently, Oregon has seen staggering caseloads and death counts, and Sullivan anticipates another big spike after Thanksgiving, which could overwhelm the state’s health systems—unless Oregonians stave off influenza infections by getting their flu shots and adhere to physical distancing guidelines even during the holidays.
“We will be seeing more and more numbers in the United States for the next three months,” he says. “We will lose another 200,000 people in the next three months in the United States unless people follow the guidance that we’ve been giving about over and over about wearing facial coverings, social distancing, keeping those Thanksgiving groups down to small numbers, and following the freeze as Governor Brown has outlined.”